Chemistry
Chemistry begins when the elements produced by astrophysical processes become capable of forming atoms, bonds, molecules, media, and reaction networks.
In the standard framework, it studies nuclei, electrons, orbitals, valence shells, bonds, molecular forms, polarity, reactions, thermodynamics, states of matter, and organic chemistry.
In Consciousness of the Real, this description is not replaced. It is reread as a series of local and collective organizations of the ( \Phi ) field, where density, coherence, spatial constraints, correlations, and media determine the accessible regimes.
The principle of caution remains central:
CdR chemistry must not announce a new chemistry as long as it does not possess operational quantities capable of producing measurable constraints.
This page therefore presents, for each stage, the recognized chemical foundations and then what CdR adds as a structural reading.
From elements to atoms
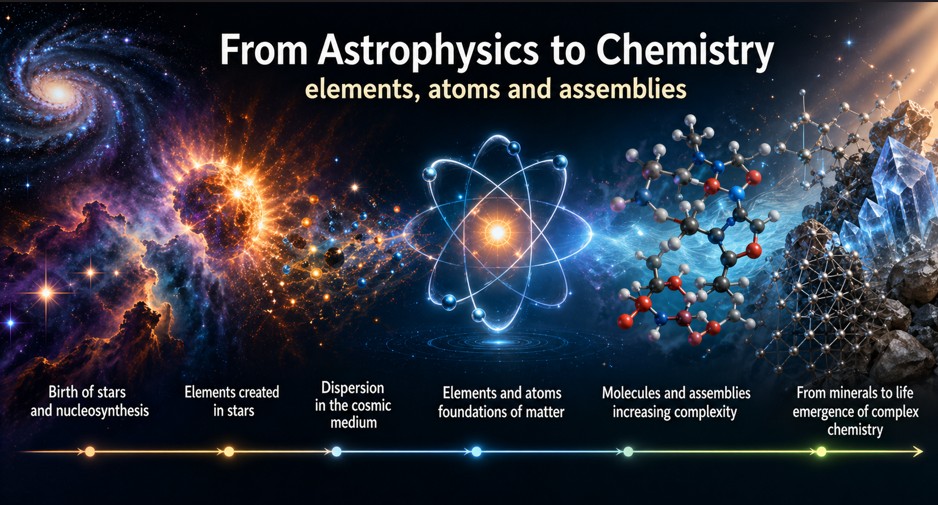
From astrophysics to chemistry — elements, atoms and assemblies
The standard basis is simple: chemical elements are not given from the outset. Nuclei form within the astrophysical history of matter: nucleosynthesis, stars, supernovae, enrichment of interstellar media, and formation of planetary systems.
Chemistry begins when these elements become available in media where they can form atoms, ions, molecules, and assemblies.
What CdR adds:
chemistry is not a rupture with the physics of matter; it is the regime where nuclei produced by cosmic processes become centers of electronic modes capable of relation.
Matter produced by astrophysics then becomes matter available for assembly.
Atoms and chemical orbitals — nuclei, valence shells and Φ modes
The standard basis is the chemical atom: a positively charged nucleus surrounded by electrons organized into levels, subshells, and orbitals. Chemical properties depend mainly on external electrons, that is, on valence shells.
Orbitals are not classical trajectories. They describe quantum states, probability distributions, energy levels, and symmetries.
What CdR adds:
an atom can be reread as a local organization where a material nucleus stabilizes stationary electronic modes.
This reading does not replace quantum mechanics. It emphasizes that the atom is already an organized structure, where electronic modes form the relational interface with other atoms.
Periodic table — chemical families and valence regimes
The standard basis is the periodic table: chemical properties repeat because external electronic configurations repeat. Alkali metals, alkaline earth metals, halogens, noble gases, transition metals, and other families express different valence regimes.
The periodic table is therefore not a simple classification. It organizes tendencies to donate, receive, share, stabilize, or delocalize electrons.
What CdR adds:
chemical families can be reread as typical regimes of external electronic coherence.
Caution remains important. CdR does not yet derive chemical periodicity or shell-closure numbers. The theoretical lock remains the link between standard orbital symmetries and the deeper grammar of the corpus.
Bonds and molecular forms
Chemical bonds — overlap of Φ modes and molecular stabilization
The standard basis is that atoms can form bonds when the bound state is more stable than the separated constituents. The main types of bonds include:
- covalent: sharing of electronic density;
- ionic: electronic transfer and electrostatic attraction;
- metallic: collective delocalization of electrons;
- coordination: asymmetric donor/acceptor sharing;
- weak interactions: polarization, dipoles, dispersion, hydrogen bonding.
What CdR adds:
a bond can be reread as a relational stabilization of valence electronic modes.
Bond types then differ by their mode of sharing or redistributing electronic density: localized sharing, transfer, collective delocalization, asymmetric coordination, or weak stabilization.
CdR does not yet derive bond energies. It proposes a structural vocabulary for linking bonding to the organization of valence modes.
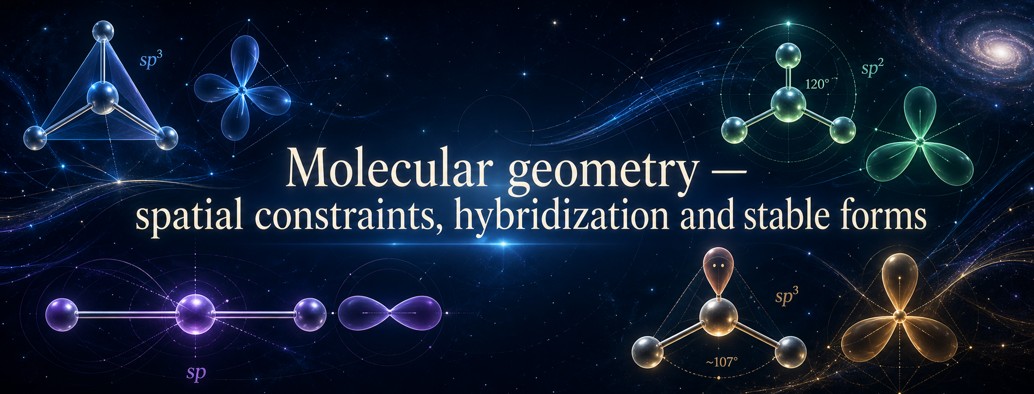
Molecular geometry — spatial constraints, hybridization and stable forms
The standard basis is that molecules have forms: linear, bent, trigonal planar, tetrahedral, pyramidal, cyclic, or more complex. These forms depend on orbitals, bonding and non-bonding pairs, electronic repulsions, hybridization, and the medium.
Models such as VSEPR, hybridization, and molecular orbitals remain indispensable.
What CdR adds:
a molecular geometry can be reread as the global spatial compatibility of valence modes.
The form of a molecule is not only a drawing: it expresses a compromise between stabilizing overlaps, repulsions, lone pairs, angle constraints, and global stability.
The first important qualitative test is the hierarchy of angles around a tetrahedral center:
[ CH_4 > NH_3 > H_2O ]
CdR does not yet derive these angles, but it identifies this terrain as a priority local test.
Polarity and intermolecular interactions — partial charges, dipoles and weak coherence
The standard basis is that molecules can have asymmetric electronic distributions. These asymmetries produce partial charges, permanent dipoles, induced dipoles, dipole-dipole interactions, dispersion forces, and hydrogen bonds.
Polarity depends both on the polarity of bonds and on global geometry. Thus, a molecule can possess polar bonds while being globally non-polar if the dipoles cancel each other.
What CdR adds:
polarity can be reread as a stable orientation of the distribution of valence electronic modes.
Weak interactions then become reversible relational stabilizations between neighboring molecules or sites.
The structural hierarchy is useful:
- hydrogen bond: strong and directional orientation;
- dipole-dipole: orientation between permanent polarization axes;
- induced dipole: polarization created by the influence of a neighbor;
- London dispersion: weak, instantaneous, and universal fluctuation.
This reading prepares liquids, solvents, membranes, proteins, and prebiotic media.
Reactions and collective regimes
Chemical reactions — reorganization paths and transition states
The standard basis is that a reaction transforms reactants into products through the breaking, formation, or reorganization of bonds. It involves a mechanism, an activation energy, a transition state, kinetics, and thermodynamics.
One must distinguish:
- thermodynamics: is the transformation favorable?
- kinetics: how fast can it occur?
- activation barrier: what critical passage must be crossed?
What CdR adds:
a reaction can be reread as a passage between two molecular organizations, through a critical and transitory configuration.
The transition state is not a stable molecule. It corresponds to a very short-lived configuration where valence modes reorganize.
CdR does not yet derive rates, equilibrium constants, or activation barriers. It provides a structural reading of the reaction passage.
Catalysis and reaction thresholds — coherence guidance and alternative paths
The standard basis is that a catalyst does not change the global thermodynamics of a reaction. It modifies the reaction path by lowering a barrier, orienting reactants, stabilizing a transition state, or providing a favorable local site.
What CdR adds:
catalysis can be reread as the opening of a lower-cost path of reorganization.
The catalyst can locally modify electronic distribution, geometry, orientation, polarization, or partial stabilization of a transition state.
This reading will become essential for prebiotic networks and enzymes, but it does not replace standard catalytic chemistry.
States of matter — collective regimes of molecular coherence
The standard basis describes states of matter through temperature, pressure, free energy, interactions, symmetries, structural order, degrees of freedom, and phase transitions.
States are not limited to the four schoolbook categories. One must also consider glasses, gels, liquid crystals, solutions, colloids, polymers, surfaces, interfaces, membranes, confined media, and complex phases.
What CdR adds:
states of matter can be reread as collective regimes of correlation between molecular modes.
The structural reading becomes:
- solid: strong structural correlations;
- liquid: dynamic local correlations;
- gas: weak and intermittent correlations;
- plasma: collective electromagnetic correlations.
Phase transitions then become changes of collective regime. CdR does not yet derive phase diagrams, but it identifies correlations, order parameters, and departures from maximum entropy as test terrains.
Chemical thermodynamics
The standard basis rests on energy, enthalpy, entropy, temperature, pressure, Gibbs free energy, and equilibrium.
At constant temperature and pressure, the central relation is:
[ \Delta G = \Delta H - T\Delta S ]
A reaction can be thermodynamically favorable without being fast. An unfavorable reaction can become possible through coupling with another favorable reaction. A chemical equilibrium is not immobility, but a dynamic compensation between reaction directions.
What CdR adds:
thermodynamic quantities can be reread as constraints on accessible chemical organizations.
The main lock is the link between entropy, free energy, and complexity (C). CdR cannot predict chemical spontaneity as long as it does not formalize this link.
A structuring track is the distinction between local complexity and extensive complexity. The proposed working relation is:
[ C_{\text{ext}} \propto S_{\max} - S ]
It remains hypothetical. It suggests that extensive complexity could measure a departure from maximum entropy under constraints, that is, a correlated organization.
Complex and prebiotic chemistry
Carbon chemistry — chains, rings, aromaticity and functional groups
The standard basis is that carbon is central in organic chemistry thanks to its tetravalence, stable bonds, (sp^3), (sp^2), and (sp) hybridizations, its capacity for catenation, its rings, aromaticity, functional groups, and chirality.
Carbon is not the only element capable of complex structures, but under known terrestrial conditions it offers a remarkable compromise between stability, flexibility, directionality, and functional diversity.
What CdR adds:
carbon can be reread as a valence center particularly suited to producing stable, flexible, and functional architectures.
Carbon chains become propagations of compatible valence regimes. Rings become geometric closures of constraints. Aromaticity becomes a case of electronic delocalization over an entire architecture. Functional groups become local modulations of polarity, reactivity, and orientation.
A structuring question remains open:
[ Z=6,\quad 2s^2 2p^2,\quad \text{tetravalence} ]
CdR must still understand why this electronic configuration produces such architectural power, and whether this fact can receive a reading within the deeper grammar of the corpus.
Prebiotic chemistry — cycles, compartments and proto-metabolisms
The standard basis of prebiotic chemistry includes water, mineral surfaces, interfaces, gradients, wet/dry cycles, compartments, autocatalysis, polymerization, reaction networks, and out-of-equilibrium systems.
Proto-life must not be presented as an automatic consequence of chemistry. One must distinguish:
- organic molecules;
- reaction networks;
- autocatalytic cycles;
- compartmentalization;
- proto-metabolism;
- replication;
- fully constituted life.
What CdR adds:
proto-organization can be reread as the progressive stabilization of persistent chemical networks in open media crossed by flows.
A more advanced prebiotic network would not merely be larger or richer in molecules. It would be more correlated, more compartmentalized, more selective, more capable of maintaining cycles and coupling reactions.
Within the working framework:
[ C_{\text{ext}} \propto S_{\max} - S ]
a more advanced proto-organization would be a network whose effective entropy departs further from the maximum entropy available under the same constraints.
This proposal remains hypothetical. It nevertheless gives CdR a qualitative criterion of its own for comparing proto-organizations.
Observable signatures of CdR chemistry — spectra, kinetics and constraints
The standard basis is that chemistry already has very precise experimental tools: spectroscopy, NMR, diffraction, neutron scattering, calorimetry, kinetics, dielectric measurements, quantum simulations, phase measurements, and correlation measurements.
What CdR adds:
these tools can serve to progressively constrain CdR quantities, provided that operational observables are defined first.
The methodological priority is clear:
define (C(\vec r)) on a simple and well-known system, then verify whether this quantity imposes a measurable constraint.
The first candidate systems are:
- water, for polarity, angles, and hydrogen-bond networks;
- benzene, for aromaticity and cyclic delocalization;
- methane, ammonia, and water, for the geometric hierarchy;
- chiral autocatalytic systems, for the amplification of homochirality.
The formula of caution for the Chemistry block is therefore:
without an operational quantity, no independent prediction.
Chirality and homochirality
The standard basis is that some molecules exist in two non-superimposable forms, like two hands. In biological systems, the selection of one chiral sign is fundamental: amino acids are predominantly of the L type, biological sugars of the D type.
What CdR adds:
chirality can be reread as spatial information carried by valence organization.
The link with the fundamental asymmetries of the corpus must remain cautious. The point is not to say that molecular chirality has already been derived. The open question is rather:
can a local chiral asymmetry be amplified, stabilized, and transmitted in a prebiotic network?
Homochirality then becomes a priority test terrain, notably through the study of chiral autocatalytic reactions.
Synthesis
The Chemistry block establishes a progressive passage:
[ \text{elements} \longrightarrow \text{atoms} \longrightarrow \text{valence} \longrightarrow \text{bonds} \longrightarrow \text{molecules} \longrightarrow \text{weak interactions} \longrightarrow \text{reactions} \longrightarrow \text{thermodynamics} \longrightarrow \text{media} \longrightarrow \text{organic chemistry} \longrightarrow \text{proto-organization} ]
CdR chemistry does not replace standard chemistry. It seeks to reformulate its main phenomena as regimes of organization, correlation, and transition.
Its main contribution, at this stage, is not a new quantitative prediction. It is an architecture of questions:
- how do electronic modes become relational?
- how do bonds stabilize common organizations?
- how do geometries constrain interactions?
- how do reactions cross thresholds?
- how do collective media stabilize correlations?
- how can prebiotic networks increase their persistence and selectivity?
- how can (C), (C(\vec r)), and (C_{\text{ext}}) be defined in a genuinely operational way?
The next step will have to transform these questions into calculable quantities and measurable tests.
Further reading
This introductory presentation is based on the technical documents of the Chemistry series:
- image100 — From astrophysics to chemistry — elements, atoms and assemblies
- image101 — Atoms and chemical orbitals — nuclei, valence shells and Φ modes
- image102 — Periodic table and chemical families — shells, valence and coherence regimes
- image103 — Chemical bonds — overlap, transfer and stabilization of electronic modes
- image104 — Molecular geometry — angles, hybridization and spatial constraints
- image105 — Polarity, dipoles and weak interactions — orientation of valence modes
- image106 — Chemical reactions — transition states, barriers and bond reorganization
- image107 — Chemical thermodynamics — free energy, entropy and equilibrium
- image108 — States of matter — phases, transitions and collective regimes
- image109 — Carbon and organic chemistry — tetravalence, chains, rings and functional groups
- image110 — Prebiotic chemistry — reaction networks, media and proto-organization
- image111 — Observable signatures of CdR chemistry — spectra, kinetics and constraints
These documents present the passage from atomic matter to molecular assemblies: chemical orbitals, bonds, forms, polarity, reactions, catalysis, states of matter, carbon chemistry, and prebiotic chemistry.